
Scientists cooled two molecules to 500 nanokelvin and forced them to enter into a delayed chemical reaction in order to capture previously elusive moments of transformation.
Chemical reactions are responsible for almost everything:breathing, cooking and digesting food, energy production and much more. Despite this, previously scientists could only theorize about how things work at a fundamental level, since the transformation occurs so quickly (in femtoseconds) that even the most precise measurements could not capture it.cameras.
In order to slow down this process and consider the course of the reaction, a team from Harvard University conducted it in ultracold conditions.
Having cooled two molecules of potassium rubidium to 500 nK theymade them literally numb. Under such conditions, the interaction was sluggish and dragged on for microseconds. Although we cannot even notice such an instant, it is millions of times slower than the normal course and enough for scientists to capture and investigate the transition phase of the transformation.
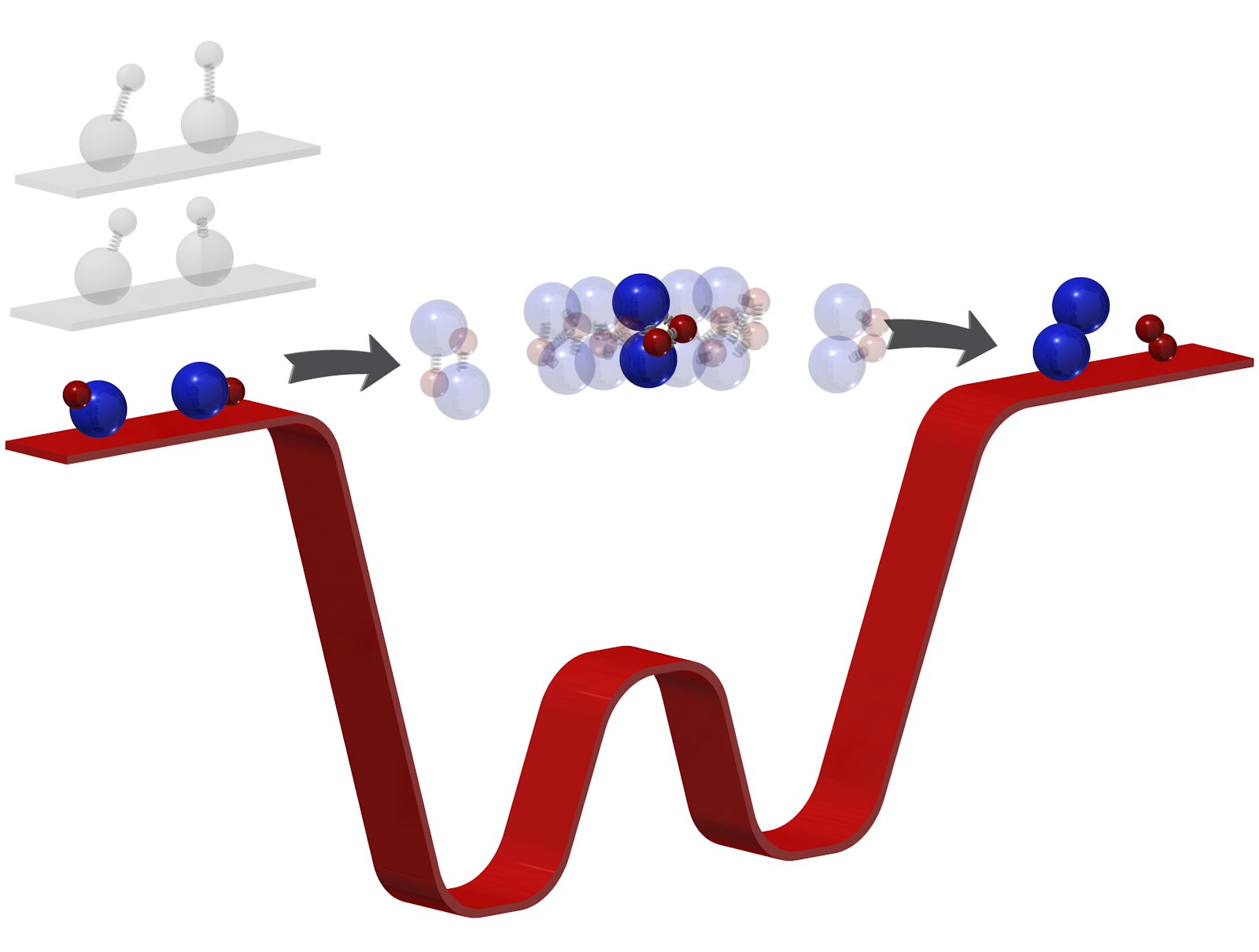
According to the team, an understanding of the fundamentals will help increase energy production efficiency, create new materials and better understand interactions in the quantum world.
Although we interact with macro objects, buttechnologies have already stepped beyond the nanolevel and are moving further. Recently, scientists have developed a model of a quantum battery that can operate without loss of energy.
</p>




